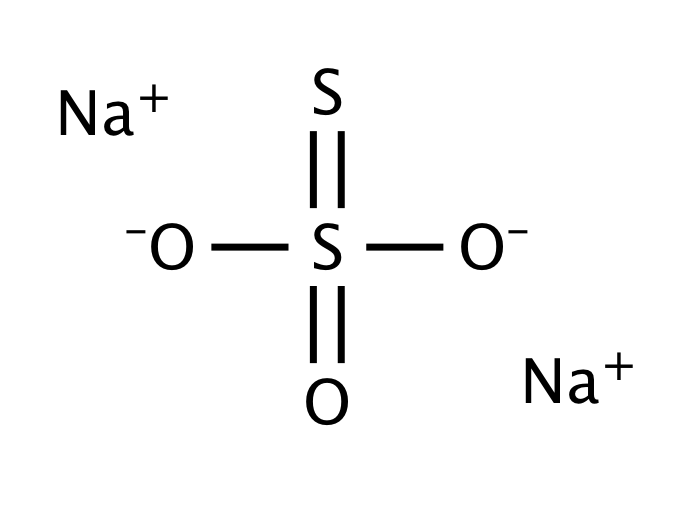Can someone please explain to me how to find the molar mass of hydrated sodium thiosulfate. I put the - Brainly.com
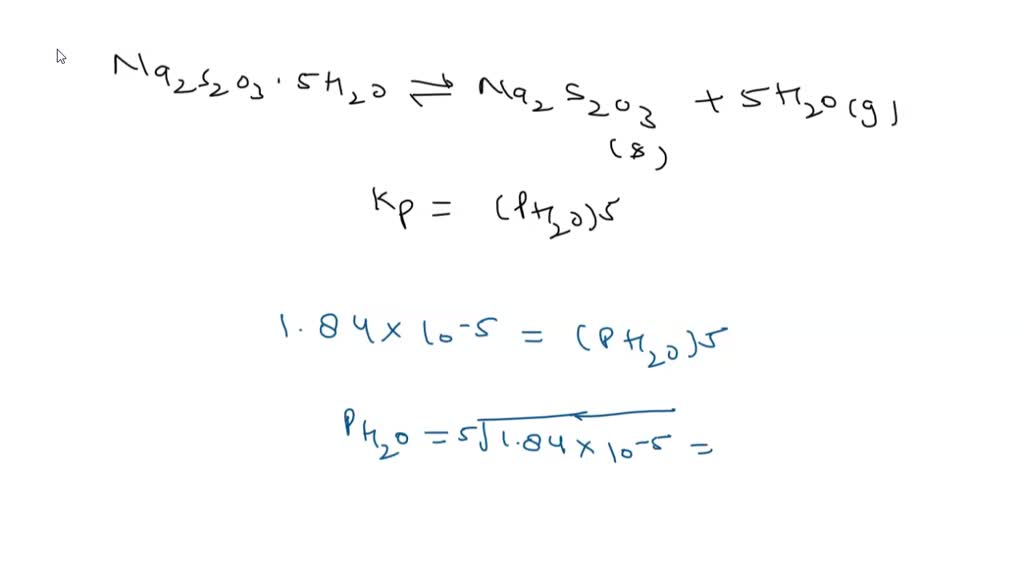
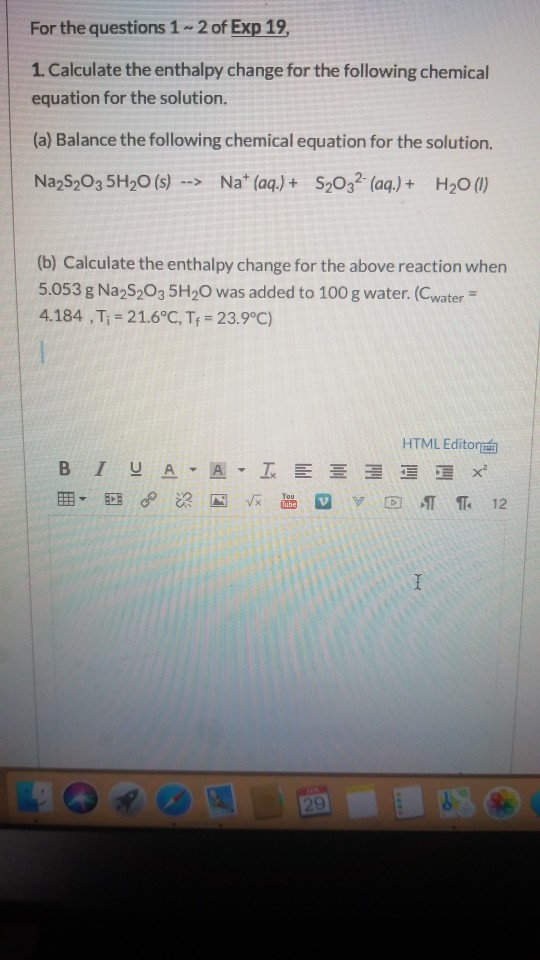
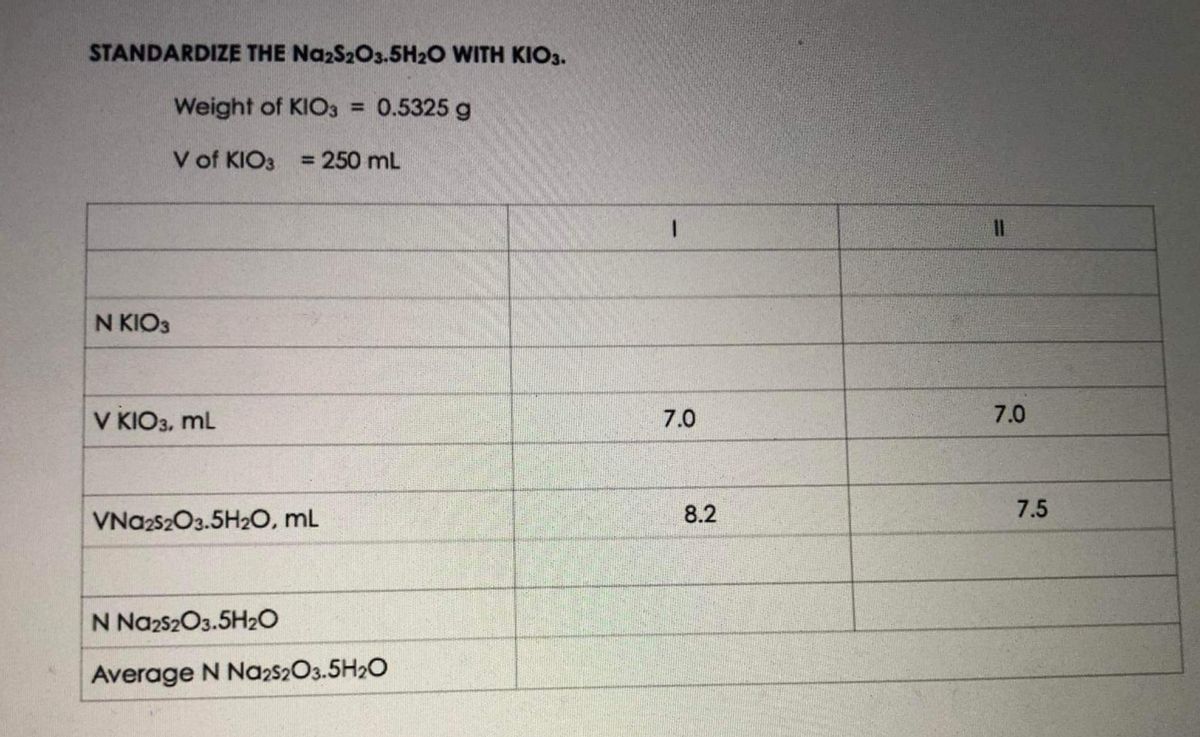
SOLVED: Sodium thiosulfate pentahydrate (FM 248.18 g ∙ mol-1) loses water when it is heated in an oven: Na2S2O3∙5H2O(s) ⇌ Na2S2O3(s) + 5H2O(g). ΔH° and ΔS° for this reaction at 25°C are

Spectrum S1502-500GM (SPFDB) Sodium Thiosulfate, Pentahydrate, Crystal, FCC, Na2S2O3.5H2O: Amazon.com: Industrial & Scientific

Uncaking Colorless Crystals Industrial Grade Na2s2o3 5H2O Sodium Thiosulfate - China Sodium Thiosulfate Anhydrous and Sodium Hyposulfite

Low Price Na2s2o3 5h2o Sodium Thiosulfate with Good Quality-Sodium thiosulfate-Tianjin Chengyuan Chemical Co., Ltd-Chengyuan

Sodium Thiosulphate 99% Min/na2s2o3.5h2o /cas No.:7772-98-7 - Buy Sodium Thiosulphate Product on Alibaba.com

SOLVED:The compound sodium thiosulfate pentahydrate, Na2 S2 O3 ·5 H2 O, is important commercially to the photography business as "hypo," because it has the ability to dissolve unreacted silver salts from photo-graphic
![Sodium Thiosulfate (Na2S2O3) [Hypo Solution Formula] - Properties, Sodium Thiosulfate Formula, Structure & Uses of Sodium Thiosulfate Sodium Thiosulfate (Na2S2O3) [Hypo Solution Formula] - Properties, Sodium Thiosulfate Formula, Structure & Uses of Sodium Thiosulfate](https://cdn1.byjus.com/wp-content/uploads/2019/03/Sodium-Thiosulfate.jpg)
Sodium Thiosulfate (Na2S2O3) [Hypo Solution Formula] - Properties, Sodium Thiosulfate Formula, Structure & Uses of Sodium Thiosulfate

Tiosulfato de sodio, sodio Hyposulfite, Na2s2o3 de 5H2O. - China Hyposulfite tiosulfato de sodio, sodio