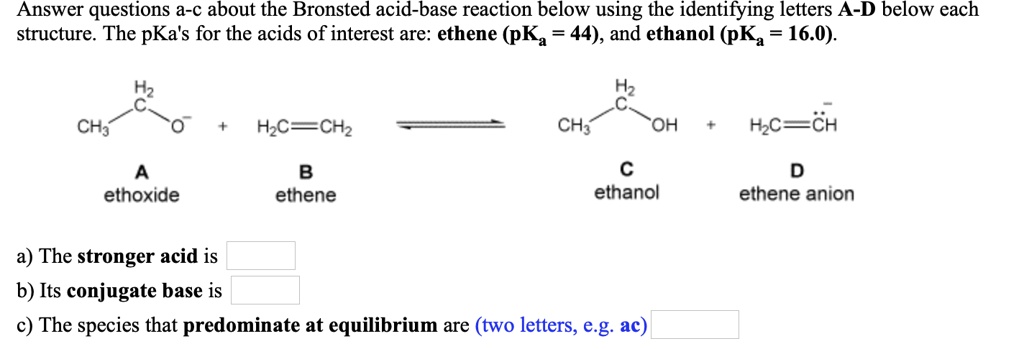
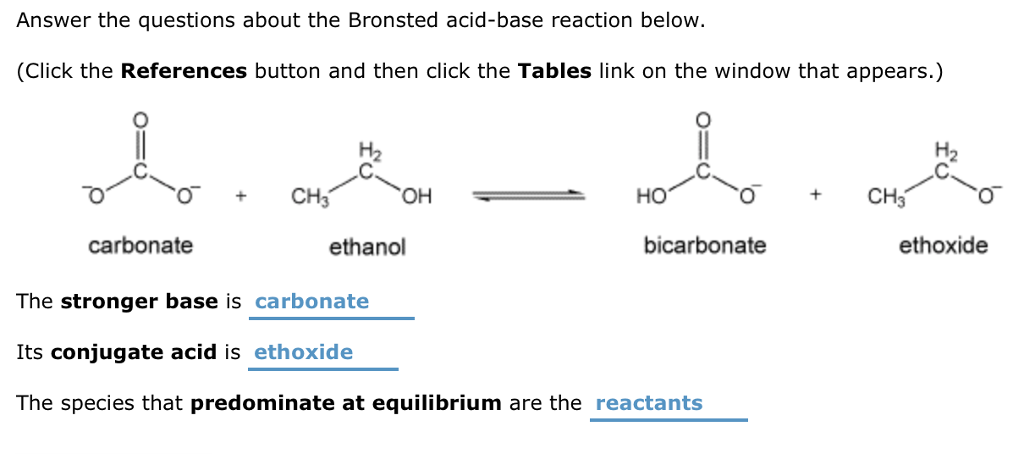
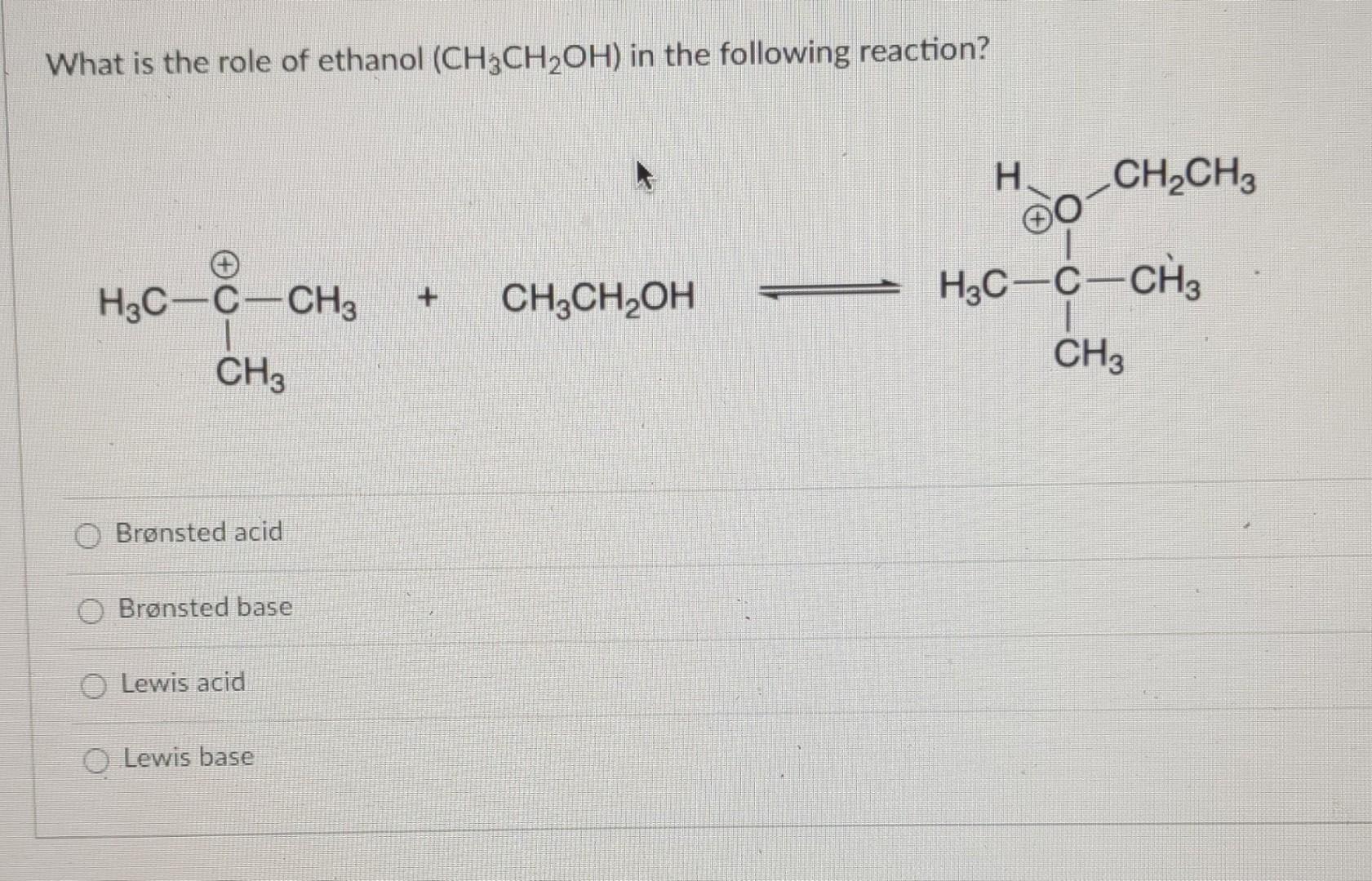
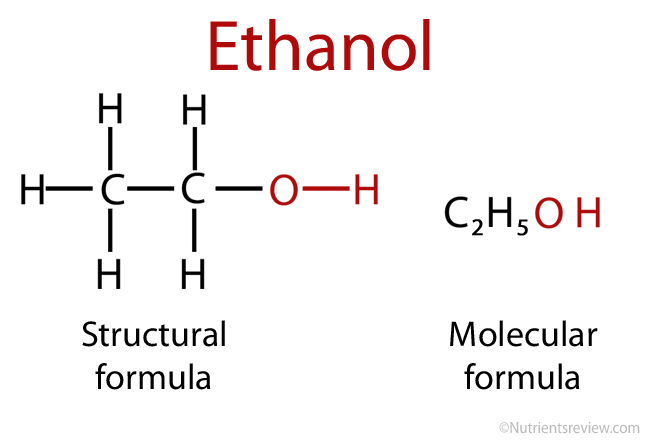
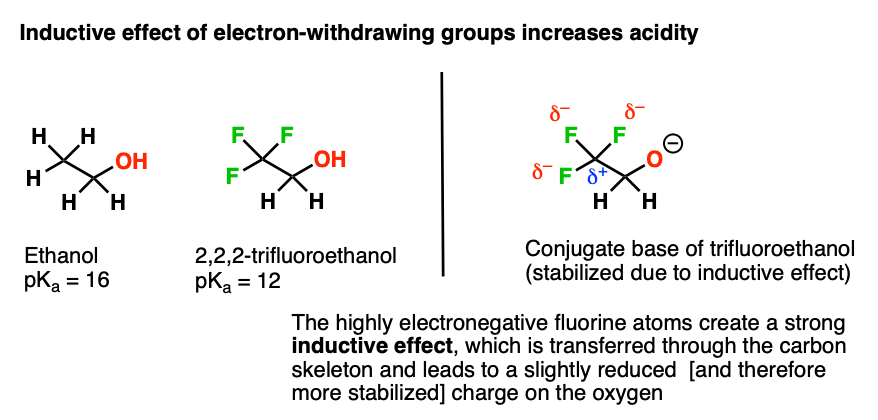
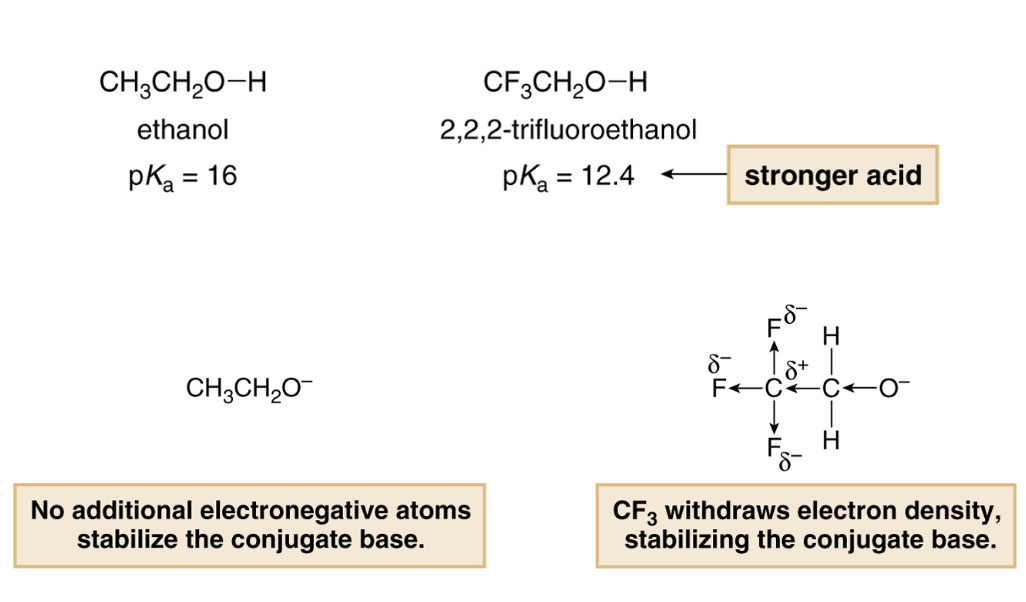
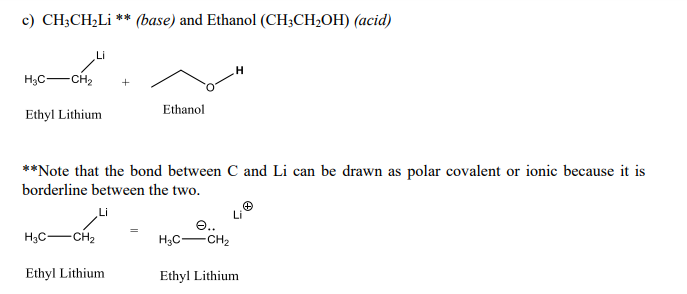
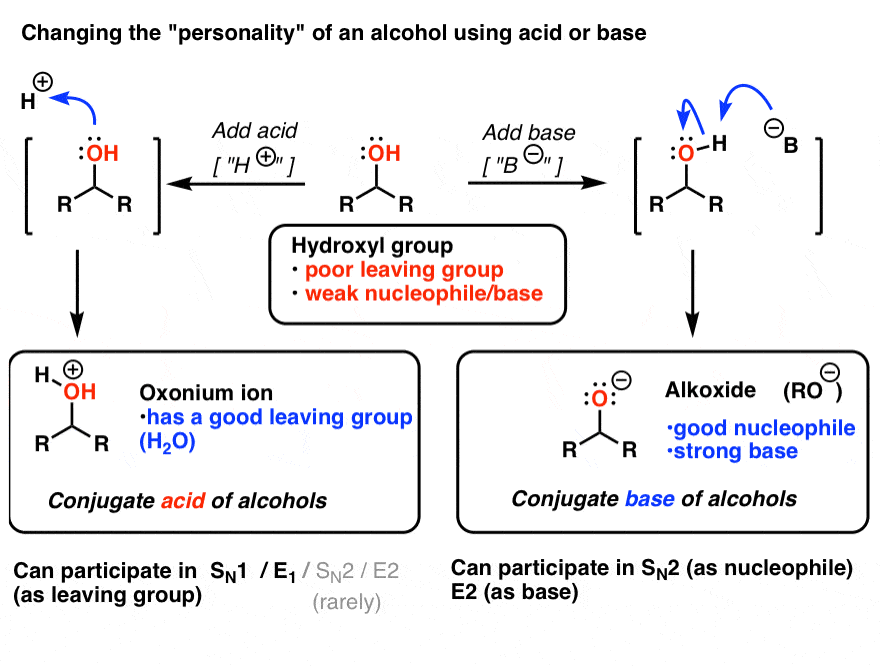
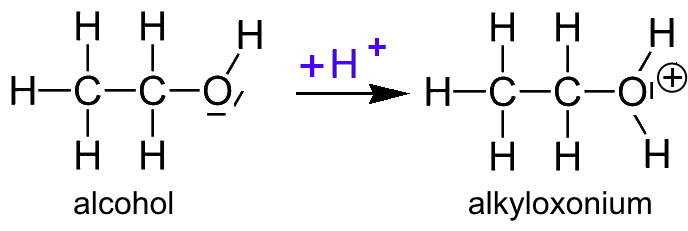1,1,1-Trichloroethanol is an acid more than 1000 times stronger than ethanol, even though both have a conjugate base where the negative charge is on an oxygen. Provide an explanation for this observation.

Mechanism of reaction between caprolactam and ethanol in the presence... | Download Scientific Diagram

Valorization of Ethanol: Ruthenium-Catalyzed Guerbet and Sequential Functionalization Processes | ACS Catalysis

Draw the Lewis structure for the conjugate base from the reaction of ethanol with a generic base. Include all lone pairs of electrons and any nonzero formal charges. | Homework.Study.com